A study on Dietary Effects on Acne and Physical Lifestyle impact
By: Ishani Mazumder
Abstract
This exploratory study investigates the potential role of dietary modulation in the management of acne vulgaris, focusing on antioxidant intake and probiotic-rich foods as mechanisms to reduce oxidative stress and systemic inflammation via the gut–skin axis. Despite limitations including a single-participant design, lack of biochemical assays, and absence of a control group, findings suggest a plausible link between improved dietary quality and acne improvement. Future research employing randomized controlled trials with larger sample sizes and direct biomarker measurements is needed to validate these mechanisms. The results contribute to the growing evidence supporting nutritional strategies as meaningful adjuncts in acne treatment. Keywords: Acne vulgaris; gut–skin axis; insulin-like growth factor-1 (IGF-1); mTORC1 signaling; oxidative stress; sebaceous gland activity; inflammatory cytokines; low-glycemic diet; antioxidants; dietary intervention
II. Introduction
Scientists are increasingly focusing on the gut-skin axis and metabolic factors. Initial research shows vitamins (A, C, and E), along with antioxidants and zinc, are great for healing inflammation.
Vitamin A is often called the “gold standard” of skin care, and it acts as a powerful cellular regulator that fundamentally changes how skin cells behave. Most topical forms must be converted into retinoic acid by the skin to bind with specific DNA receptors.
Vitamin C, known as Ascorbic acid, is a potent antioxidant and serves as a repair agent for the skin, unlike Vitamin A. Vitamin C neutralizes environmental damage and inhibits pigment production.
Vitamin E (tocopherol) is a fat-soluble antioxidant that scavenges free radicals produced by UV radiation and pollutants, which prevents early aging and cell damage.
Vitamins mentioned are bases for healthier skin as it provides collagen production (done by Vitamin C), cell regeneration (Vitamin A), and wound healing (Vitamin C and K). These three act as “biological building blocks” and “bodyguards” for the body’s largest organ. Through the intake of bioactive molecules such as vitamins, antioxidants, fatty acids, and minerals.
Beta-carotene, for example, for neutralizing reactive oxygen species (ROS). ROS are unstable molecules produced during normal metabolism and increased UV exposure; they can damage cellular lipids, proteins, and DNA within skin cells. Excess oxidative stress can stimulate inflammatory pathways and increase sebum production, both of which contribute to acne development. Diets high in refined carbohydrates can also elevate insulin and insulin-like growth factor-1 (IGF-I), which increases androgen signaling and stimulates sebaceous glands too produce excess oil.
Water hydration for skin structure and function at a cellular level. Molecules contribute to skin elasticity, enzyme activity, and the transport of nutrients across cell membranes. Inadequate hydration can impair the stratum corneum barrier, leading to trans epidermal water loss. To compensate for dryness, sebaceous glands may increase lipid secretion, resulting in clogged pores and creating an environment favorable for acne-causing bacteria.
Post-scientific research increasingly confirms that acne is not a solely genetic condition but rather influenced by the “exposome,” the sum of dietary and lifestyle factors. Healthy skin is vital as the body’s largest organ, acting as a crucial barrier against infection. Also. serves as a hydration supplier and protects from UV damage. In essence, healthy skin in your body is your first line of defense and a window to your internal health. Which is why taking care of it is important. Lifestyle factors, such as having a good diet and getting good sleep. By fulfilling these factors, one can experiences few disease infections and an increase in antioxidant benefits. Diet, hydration, and skin exposure can play a significant role in acne development and skin health, understanding these outcomes can help prevent skin conditions. Background Pre-research
This study was conducted as a structured literature review. The relationship between nutrition, sun exposure, and acne development at the molecular and biochemical levels. The review focused on hormonal regulation and microbial interactions involved in acne pathogenesis.
An in-depth search of electronic databases, including PubMed, Google Scholar, ScienceDirect, and articles published in JAMA Dermatology, was conducted. The search included studies published between 2013 and 2024. Keywords and Boolean operators were used to identify relevant studies, including combinations such as “acne vulgaris AND glycemic index,” “IGF-1 AND sebaceous glands,” “vitamin D AND acne inflammation,” “omega-3 fatty acids AND skin inflammation,” and “UV radiation AND acne pathogenesis.” Reference lists of selected articles were also manually reviewed to identify additional relevant sources.
Studies were included if they were peer-reviewed journal articles involving human participants and examined biological mechanisms related to acne. Eligible study designs included randomized controlled trials, cohort studies, cross-sectional studies, systematic reviews, and meta-analyses. Articles were required to assess measurable outcomes such as acne severity scores, serum hormone levels (e.g., insulin or insulin-like growth factor-1), inflammatory cytokines, oxidative stress markers, or sebaceous gland activity. Studies focusing merely on cosmetic treatments without discussion of underlying molecular mechanisms were excluded. Non-English publications and case reports without a mechanistic analysis were also excluded.
Relevant data were extracted from each study, including sample size and participant demographics, type of dietary or environmental exposure, measured biological markers, and reported findings. The collected data were analyzed qualitatively by categorizing studies according to major biological pathways, including hormonal signaling (insulin, IGF-1, and mTORC1 activation), inflammatory mediators (such as interleukin-1β and tumor necrosis factor-α), sebaceous gland lipid production, microbial activity involving Cuti bacterium acnes, and vitamin D metabolism following UV exposure. Patterns and consistencies across studies were evaluated to determine the strength of evidence supporting the association between lifestyle factors and acne pathophysiology.
III. Materials and Methods
A 30-day self-monitored dietary intervention evaluating the impact of a low-glycemic, antioxidant-rich lunch on acne severity. In addition to the literature review, a 30-day self-conducted dietary intervention was performed to explore the short-term effects of a low-glycemic, antioxidant-rich lunch on acne severity. The intervention consisted of replacing a standard lunch meal with a fruit-based meal including almonds, dried cranberries (craisins), blueberries, cantaloupe, strawberries, banana, oranges, and plain Greek yogurt. The selected foods were chosen due to their antioxidant content (vitamin C, polyphenols), fiber content, and low to moderate glycemic load, as well as the probiotic and protein content of Greek yogurt.
The participant maintained all other aspects of diet and lifestyle as consistently as possible during the 30 days. No new skincare products or medications were introduced during the study. Acne severity was monitored weekly using lesion counts (inflammatory and non-inflammatory lesions) and photographic documentation under consistent lighting conditions. Subjective measures, including skin oiliness and inflammation, were also recorded in a daily log.
The primary variables assessed were changes in lesion count, visible inflammation, and overall skin clarity over the 30 days. While the study was limited to a single participant and lacked a control group, it served as a preliminary observational model to compare real-world findings with mechanisms identified in the literature review.
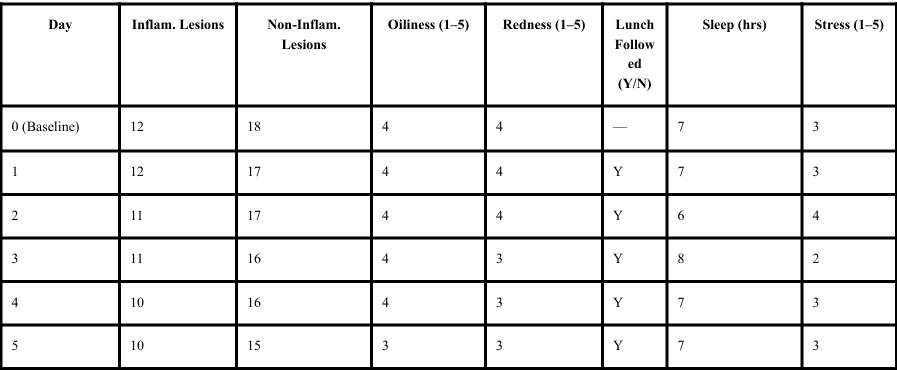
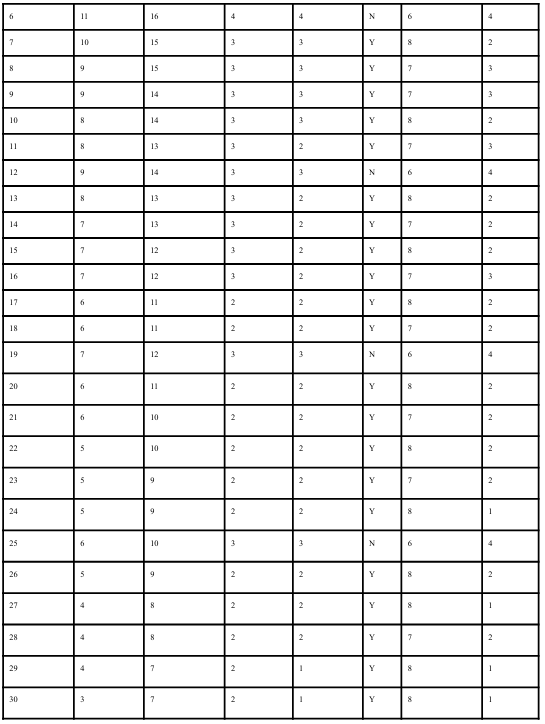
Variables
Sebaceous activity was assessed using a subjective oiliness scale ranging from 1 (very dry) to 5 (very oily), evaluated at approximately the same time each day. Cutaneous erythema was measured using a standardized redness scale ranging from 1 (no visible redness) to 5 (severe inflammation). Secondary variables included dietary adherence (recorded as yes/no), sleep duration (hours per night), and perceived stress level measured on a 1–5 scale. These secondary variables were included to account for potential confounding factors known to influence hormonal signaling, sebaceous gland activity, and inflammatory response.
Results
Post acne Inflammation Photo (After the study was conducted)
Quantitative evaluation of the 30-day dietary intervention revealed a sustained reduction in acne lesion burden and associated inflammatory characteristics. Baseline assessment demonstrated 12 inflammatory lesions and 18 non-inflammatory lesions, accompanied by elevated sebaceous activity (oiliness score: 4/5) and pronounced cutaneous erythema (4/5). By Day 30, inflammatory lesions declined to 3 and non-inflammatory lesions to 7, corresponding to a 75% reduction in inflammatory lesions and a 61% reduction in comedonal lesions relative to baseline.
The downward trajectory in lesion counts became more pronounced after Day 14, suggesting a delayed but progressive physiological response. Weekly mean oiliness scores decreased from approximately 4.0 during Week 1 to 2.0 by Week 4, indicating a measurable reduction in sebaceous gland activity. Because sebaceous lipid production is influenced by insulin and insulin-like growth factor-1 (IGF-1) signaling, this reduction may reflect improved glycemic stability and decreased activation of the mTORC1 (mechanistic target of rapamycin complex 1) pathway, which is known to stimulate sebocyte proliferation and lipogenesis. Similarly, erythema scores declined steadily from 4/5 to 1/5 over the intervention period, suggesting attenuation of cutaneous inflammation.
This improvement is consistent with reduced expression of pro-inflammatory cytokines such as interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α), which play central roles in acne pathogenesis. The antioxidant-rich composition of the intervention meal — including vitamin C, polyphenols, vitamin E, and flavonoids — may have contributed to decreased reactive oxygen species (ROS) production and oxidative stress within pilosebaceous units. Transient increases in lesion count and sebaceous activity were observed on Days 6, 12, 19, and 25. These fluctuations coincided with missed dietary adherence, elevated perceived stress levels, and reduced sleep duration. Acute stress is known to increase cortisol secretion, which can indirectly enhance sebaceous activity and inflammatory signaling. However, following the resumption of consistent dietary intake and improved sleep, lesion counts returned to the overall downward trend.
Collectively, these findings suggest that sustained consumption of a low-glycemic, fiber-rich, antioxidant-dense dietary pattern may be associated with reduced sebaceous lipogenesis, modulation of insulin/IGF-1 signaling, decreased mTORC1 activation, and attenuation of inflammatory cytokine activity. While the absence of biochemical assays prevents direct measurement of these molecular markers, the observed clinical trends align with established mechanistic pathways described in dermatologic literature. Given the single-participant design and lack of a control group, these findings remain exploratory and hypothesis-generating rather than confirmatory.
Discussion
The findings of this 30-day dietary intervention suggest a potential association between consistent consumption of a low-glycemic, antioxidant-rich lunch and reductions in acne lesion burden, sebaceous activity, and visible inflammation. The progressive decline in inflammatory and non-inflammatory lesions observed over the intervention period aligns with established mechanistic models of acne pathogenesis involving insulin signaling, IGF-1 activity, and mTORC1 pathway activation. Acne development is strongly influenced by insulin and insulin-like growth factor-1 (IGF-1), both of which promote sebocyte proliferation and lipogenesis through activation of the mechanistic target of rapamycin complex 1 (mTORC1). Diets characterized by high glycemic load can induce hyperinsulinemia, thereby upregulating IGF-1 and stimulating excessive sebum production. The fruit-based lunch intervention, while containing natural sugars, was rich in fiber and paired with protein and healthy fats from Greek yogurt and almonds, likely moderating postprandial glucose spikes. Improved glycemic stability may therefore have contributed to reduced sebaceous output, as reflected by the decline in oiliness scores from 4/5 to 2/5 over the study period. In addition to hormonal modulation, oxidative stress plays a critical role in acne inflammation. Reactive oxygen species (ROS) contribute to lipid peroxidation within the pilosebaceous unit, amplifying inflammatory cascades and cytokine release. The intervention meal included high concentrations of vitamin C (oranges, strawberries), polyphenols (blueberries), carotenoids (cantaloupe), and vitamin E (almonds), all of which possess antioxidant properties.
The observed reduction in erythema and inflammatory lesions may therefore reflect attenuation of oxidative stress and decreased expression of pro-inflammatory cytokines such as interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α). The inclusion of Greek yogurt also introduces a potential gut–skin axis mechanism. Emerging research suggests that modulation of gut microbiota composition may influence systemic inflammation and immune regulation. Probiotic-containing foods may help reduce systemic inflammatory tone, potentially influencing the cutaneous immune response associated with Cuti bacterium acnes colonization. Although microbiome composition was not directly measured in this study, the consistent improvement observed after two weeks may correspond to gradual microbial adaptation. Notably, temporary increases in lesion count were observed during periods of elevated stress and reduced sleep. Psychological stress is known to increase cortisol secretion, which can enhance sebaceous gland activity and inflammatory signaling pathways. These fluctuations highlight the multifactorial nature of acne and suggest that dietary modulation may interact with neuroendocrine influences. Despite these promising trends, several limitations must be acknowledged. The single-subject design limits generalizability, and the absence of biochemical assays prevents direct measurement of insulin levels, IGF-1 concentration, cytokine expression, or mTORC1 activity. Additionally, the lack of a control group precludes definitive causal conclusions. The improvements observed may partially reflect natural acne variability or placebo effects. Future research involving randomized controlled designs, larger sample sizes, and molecular biomarker analysis would be necessary to confirm these findings.
Conclusion
The observed clinical trends are biologically consistent with established mechanisms of acne pathogenesis. Improved glycemic regulation may reduce insulin and insulin-like growth factor-1 (IGF-1) signaling, thereby attenuating mTORC1 pathway activation and sebaceous lipogenesis. Additionally, increased antioxidant intake may mitigate oxidative stress within the pilosebaceous unit, potentially decreasing lipid peroxidation and pro-inflammatory cytokine activity. The inclusion of probiotic-rich foods may further contribute to the modulation of the gut–skin axis and systemic inflammatory tone.
While the findings support a plausible mechanistic link between dietary quality and acne improvement, important limitations must be acknowledged. The single-participant design, absence of biochemical assays, and lack of a control group limit the ability to establish causality. Therefore, the results should be interpreted as exploratory and hypothesis-generating rather than definitive evidence of therapeutic efficacy.
Future research employing randomized controlled methodologies, larger sample populations, and direct measurement of hormonal and inflammatory biomarkers would provide stronger validation of the proposed mechanisms. Nevertheless, this study reinforces the growing body of evidence suggesting that nutritional modulation may represent a meaningful adjunct strategy in the management of acne vulgaris.
References
Bowe, W. P., & Logan, A. C. (2011). Acne vulgaris, probiotics and the gut–brain–skin axis: From anecdote to translational medicine. Gut Pathogens, 3(1), 1. https://doi.org/10.1186/1757-4749-3-1https://gutpathogens.biomedcentral.com/articles/10.1186/1757-4749-3-1
Dreno, B., Pécastaings, S., Corvec, S., Veraldi, S., Khammari, A., & Roques, C. (2018). Cutibacterium acnes (Propionibacterium acnes) and acne vulgaris: A brief look at the latest updates. Journal of the European Academy of Dermatology and Venereology, 32(Suppl 2), 5–14. https://doi.org/10.1111/jdv.15043 https://onlinelibrary.wiley.com/doi/10.1111/jdv.15043
Katta, R., & Desai, S. P. (2014). Diet and dermatology: The role of dietary intervention in skin disease. Journal of Clinical and Aesthetic Dermatology, 7(7), 46–51. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4106357/
Melnik, B. C. (2015). Linking diet to acne metabolomics, inflammation, and comedogenesis: An update. Clinical, Cosmetic and Investigational Dermatology, 8, 371–388. https://doi.org/10.2147/CCID.S69135 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4565837/
Melnik, B. C., John, S. M., & Schmitz, G. (2013). Over-stimulation of insulin/IGF-1 signaling by Western diet may promote diseases of civilization: Lessons from acne. Dermato-Endocrinology, 5(1), 1–6. https://doi.org/10.4161/derm.22665 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3583891/
Ozuguz, P., Dogruk Kacar, S., Ekiz, O., Takci, Z., & Bas, Y. (2014). Evaluation of serum vitamins A and E and zinc levels according to the severity of acne vulgaris. Cutaneous and Ocular Toxicology, 33(2), 99–102. https://doi.org/10.3109/15569527.2013.808655 https://pubmed.ncbi.nlm.nih.gov/23865475/
Smith, R. N., Mann, N. J., Braue, A., Mäkeläinen, H., & Varigos, G. A. (2007). The effect of a low-glycemic-load diet on acne vulgaris and the fatty acid composition of skin surface triglycerides. Journal of the American Academy of Dermatology, 57(2), 247–256. https://doi.org/10.1016/j.jaad.2007.01.046 https://pubmed.ncbi.nlm.nih.gov/17617492/
Zaenglein, A. L., et al. (2016). Guidelines of care for the management of acne vulgaris. Journal of the American Academy of Dermatology, 74(5), 945–973. https://doi.org/10.1016/j.jaad.2015.12.037 https://pubmed.ncbi.nlm.nih.gov/26897386/
